Sports
Sports event coverage and updates

Written by William Farrand, Senior Research Scientist, Space Science Institute Earth planning date: Friday, March 6, 2026 Curiosity is in the last stage of its exploration of the spiderweb-like boxwor...
March 11, 2026
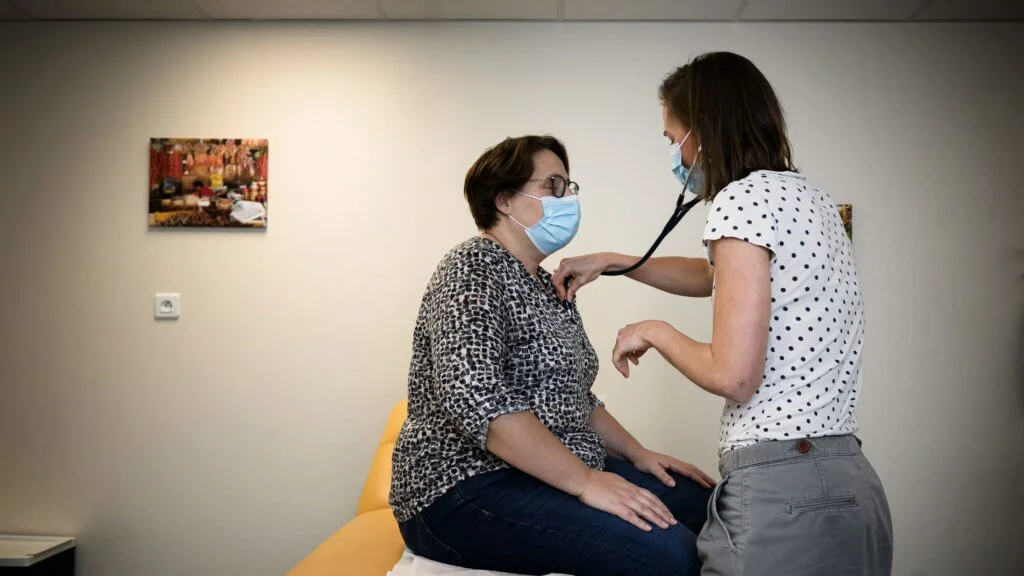
In medical school, a student writes, she has learned to react first, focus on prevention later.
March 11, 2026

Trump raised concerns that the players would “most likely be killed” if they had to return to Iran.
March 09, 2026

CAIR releases annual report, warning discrimination is likely to continue as Iran war sparks a new wave of hate speech.
March 10, 2026

Party members decry 'disturbing' lack of clarity following latest classified briefing on war justification and aims.
March 10, 2026

Australia's home affairs minister says a total of seven members of the Iranian women's team have been granted asylum.
March 11, 2026

Lawmakers say the White House has not clearly explained why the US entered the conflict, or how long it may last.
March 11, 2026

A fleet of NASA missions has likely uncovered a collision between two ultradense stars in a tiny galaxy buried in a huge stream of gas. Astronomers have never seen this type of explosive event in an e...
March 10, 2026
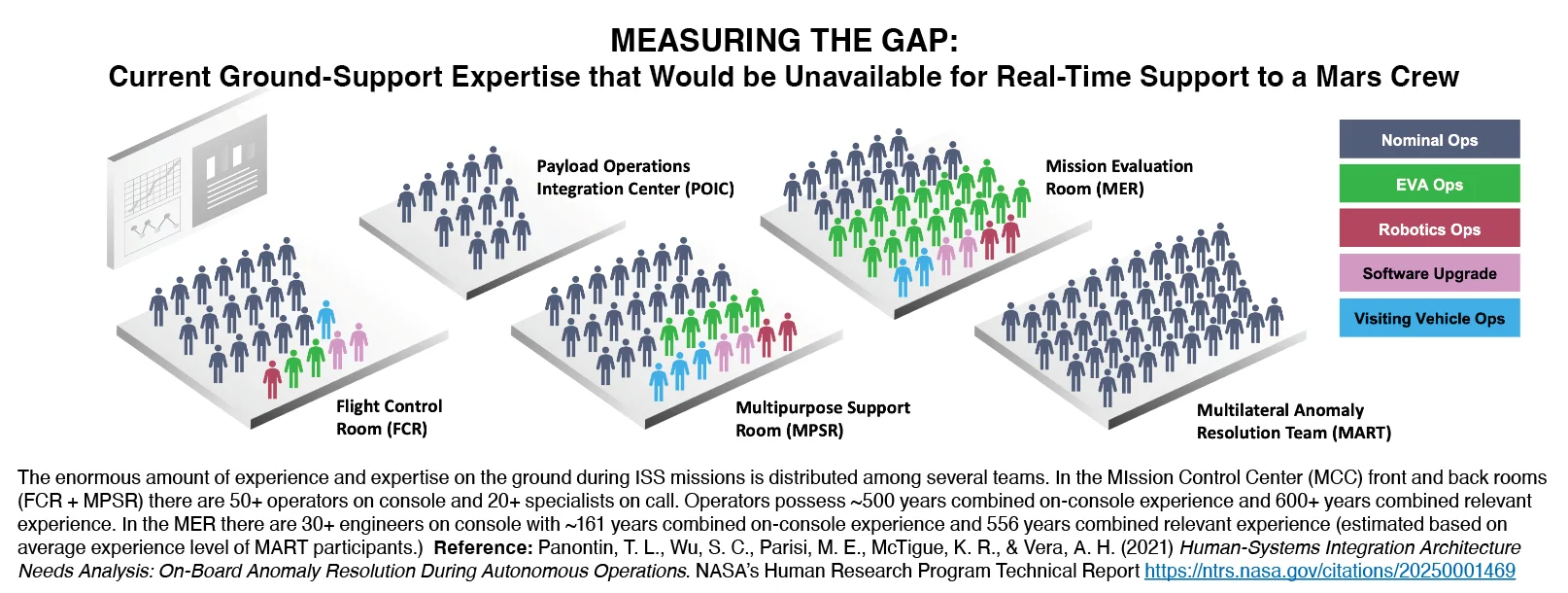
This article is from the 2025 Technical Update. The human factors TDT looks for and creates opportunities to influence design to leverage human strengths and to protect people and missions. The human ...
March 10, 2026

Ukraine has built low-cost drones to counter Russian attacks with imported Iranian Shahed drones.
March 09, 2026

Ministry of National Defence says no casualties or damage after missile shot down over southern city of Gaziantep.
March 09, 2026

Australian PM says the Iranian team members ‘asked for assistance and had received it and were safely located’.
March 09, 2026

As strikes continue across Iran, US President Trump has articulated shifting goals. But how likely are they?
March 09, 2026

**Vinay Prasad's Controversial Stance on Rare Disease Therapeutics: A Regulatory Crossroads**
FDA oncology division leader Vinay Prasad has emerged as a polarizing figure in rare disease drug policy, repeatedly challenging approval pathways that patient advocates and biopharma stakeholders consider critical lifelines. His decisions have sparked intense debate across the drug development ecosystem — raising fundamental questions about the balance between accelerated access, clinical evidence standards, and regulatory risk tolerance in orphan disease therapeutics.
For rare disease patients — where treatment options are scarce and trial populations inherently small — Prasad's evidence-first posture carries outsized consequences, potentially reshaping how sponsors design pivotal studies and how payers ultimately assess value in ultra-rare indications.
March 06, 2026

Behavioral and developmental pediatrician Lawrence Diller has observed a growing trend: parents actively seeking autism diagnoses for their children. Drawing from his clinical experience, Diller offers a nuanced perspective on the psychological and systemic drivers behind this phenomenon — from expanded diagnostic criteria and increased awareness to the practical reality that an autism spectrum disorder (ASD) label often unlocks access to educational resources, therapeutic services, and insurance coverage that would otherwise remain out of reach. In an era where neurodevelopmental conditions are increasingly destigmatized and advocacy communities thrive online, the diagnosis itself has become a gateway — not a stigma — prompting families to pursue formal evaluation with unprecedented urgency. Diller's insights challenge clinicians to examine how societal pressures, resource scarcity, and evolving cultural narratives around neurodiversity are reshaping the diagnostic landscape in pediatric medicine.
March 09, 2026

# Flawed Drug Screening in Healthcare: Why Hospitals Are Rethinking Mandatory Testing Protocols
Hospitals across the U.S. have long subjected new mothers to routine urine drug screenings — often without informed consent — then reported positive results to child protective services based on tests with documented high false-positive rates. The consequences have been devastating: families separated, parental rights challenged, and marginalized communities disproportionately targeted, all driven by unreliable immunoassay screens that can flag common medications, poppy seeds, and over-the-counter compounds as illicit substances.
Now, a growing coalition of physicians, bioethicists, and patient advocates is challenging this standard of care. The core argument is both clinical and ethical: when a diagnostic tool's error rate is high enough to destroy families, its routine deployment constitutes harm — a direct violation of medical primum non nocere principles.
Forward-thinking health systems are responding by replacing blanket screening mandates with risk-stratified, consent-based protocols, while simultaneously demanding confirmatory GC-MS testing before any mandated reporting occurs. This shift reflects broader healthcare trends toward **evidence-based policy reform**, **health equity**, and **patient data rights** — recognizing that algorithmic or procedural shortcuts in clinical workflows carry real human costs.
The takeaway for healthcare administrators and policymakers: operational efficiency cannot override diagnostic accuracy when the downstream impact is family dissolution. Reforming drug testing protocols isn't just compassionate medicine — it's sound risk management.
April 02, 2025

# Facial Recognition as Due Process: When Biometrics Become a Citizen's Last Defense
A U.S. citizen's freedom hinged on a facial recognition scan. Jesus Gutiérrez, wrongly detained by immigration enforcement agents, was only released after ICE's biometric identification technology confirmed his citizenship status — a striking case that exposes both the growing role of AI-powered identity verification in federal law enforcement and the systemic vulnerabilities that make such technology necessary in the first place.
The incident raises critical questions at the intersection of civil liberties, algorithmic accountability, and immigration enforcement policy. While facial recognition ultimately exonerated Gutiérrez, the episode underscores a troubling paradox: biometric surveillance tools simultaneously represent a potential safeguard for citizens *and* a significant threat to privacy rights and due process when deployed without robust oversight frameworks.
For tech industry professionals, this case serves as a real-world stress test of computer vision systems operating in high-stakes, irreversible decision environments — precisely the scenarios where algorithmic bias, false positive rates, and chain-of-custody data integrity carry life-altering consequences. It also accelerates an urgent policy conversation around **ethical AI deployment standards**, explainability requirements, and the legal liability gap that currently exists when automated systems influence law enforcement outcomes.
As federal agencies scale biometric infrastructure, the Gutiérrez case isn't just a human interest story — it's a governance benchmark.
December 13, 2025







